|
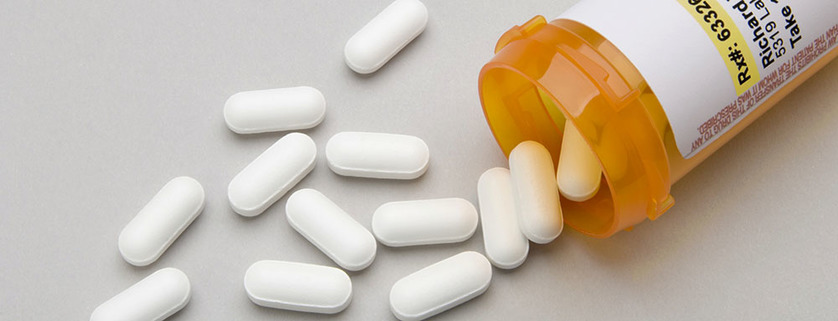
Pharmacy chain CVS Health Corp. has agreed to pay $450,000 to resolve the United States’ allegations that several of its Rhode Island stores violated the federal Controlled Substances Act by filling invalid prescriptions and maintaining deficient records, announced United States Attorney Peter F. Neronha and Michael J. Ferguson, Special Agent in Charge of the Drug Enforcement Administration’s New England Field Division. The settlement caps an investigation by the United States Attorney’s Office for the District of Rhode Island and the DEA Office of Diversion Control of CVS’ conduct at its Rhode Island retail pharmacy locations in filling prescriptions for various controlled substances with a high potential for abuse. Under the Controlled Substances Act and its implementing regulations, substances such as painkillers and opioids can only be prescribed for legitimate medical purposes by a physician. The law, however, also places a “corresponding responsibility” on the pharmacist filling the prescription to ensure that the prescription is valid and legal, and that that the prescriber has the legal authority to prescribe the drug. The Act also imposes a number of recordkeeping requirements.
In this case, the United States alleged that CVS retail pharmacies in Rhode Island filled a number of forged prescriptions with invalid DEA numbers, and filled multiple prescriptions written by psychiatric nurse practitioners for the opioid painkiller hydrocodone, despite the fact that these practitioners were not legally permitted to prescribe these drugs. Additionally, the government alleged that CVS had recordkeeping deficiencies. Under the terms of a civil agreement, CVS will pay a civil penalty of $450,000 for its alleged conduct in the State of Rhode Island. CVS, while acknowledging that its pharmacists bear a corresponding responsibility under the Controlled Substances Act, has denied wrongdoing in this case. “The diversion and misuse of prescription painkillers are a public health crisis in the State of Rhode Island,” commented U.S. Attorney Peter F. Neronha. “Even in cases where there is no evidence of abuse or nefarious intent, this Office remains committed to using all the legal tools at our disposal to ensure that everyone in the chain of controlled substance distribution -- from physicians to pharmacists -- abides by the strict requirements of the law.” “DEA registrants are responsible to handle controlled substances in compliance with the Controlled Substance Act,” said Special Agent in Charge Michael J. Ferguson. “Our obligation is to improve public safety and public health, and we are committed to working with our law enforcement and regulatory partners nationwide to ensure that these rules and regulations are followed.” This matter was litigated by Assistant U.S. Attorneys Zachary A. Cunha and Bethany N. Wong. |
WBOB
|
Search For Your Favorite WBOB Author,
or BobCast
990WBOB
An Independent Media Outlet.
The views opinions and thoughts expressed do not reflect those of 990WBOB, its management or its staff. All Rights Reserved 990WBOB.com 2007-2020
Contact WBOB HERE
An Independent Media Outlet.
The views opinions and thoughts expressed do not reflect those of 990WBOB, its management or its staff. All Rights Reserved 990WBOB.com 2007-2020
Contact WBOB HERE